
This client is a multinational oncology contract manufacturer. Their production was ceased at the instruction of US-FDA to reflect further changes to the Validation Approach or to the scope of the equipment, facility, or utilities. PharmEng provided various services to restart production for four manufacturing facilities at the client’s site, which supported multiple lines. The[…..] […]
Read More…

PharmEng was retained to develop the Basis of Design for an IPA facility in Minnesota. Upon completion of the construction, PharmEng was later retained to provide a gap analysis of the completed construction project for compliance with Health Canada and FDA Animal Health GMPs in 2009. Responsibilities: Basis of Design Functional and Space Programming Equipment[…..] […]
Read More…
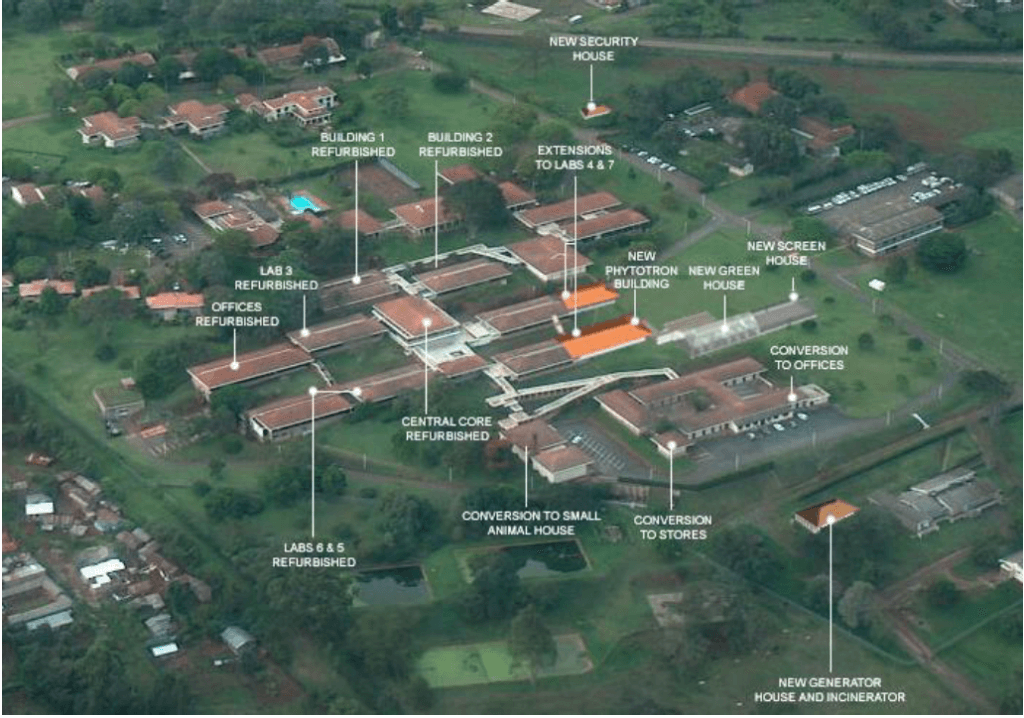
This client facilitates research of livestock and plant science with the objective of reducing poverty in Central and Eastern Africa. PharmEng was engaged to provide technical expertise and assess the facility’s environmental impact. PharmEng utilized its North American design experiences to solve complex functional flows in animal care areas and BSL3 Laboratories. Responsibilities: Functional Program[…..] […]
Read More…

Canadian Phytopharmaceuticals Corp. (CPC) is a leading custom manufacturer and private labeler of top quality botanical herbal extracts and health food products PharmEng reviewed CPC’s conceptual design and recommended modifications to meet Health Canada – Natural Health Product Directorate’s GMPs. CPC has obtained their NHP-GMP license and is now fully operational. Responsibilities: GMP Plan Review[…..] […]
Read More…










